Alternative to Statin Titration
Double statin dose doesn't equal double efficacy1
Consider Welchol when higher doses of a statin are no longer an option.
Welchol can be an alternative to high-dose statin titration therapy2-7
Welchol provides added LDL-C efficacy, without systemic absorption
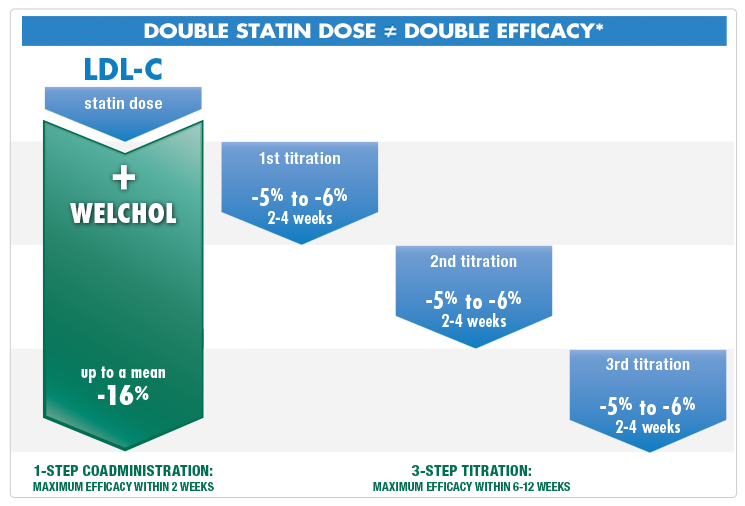
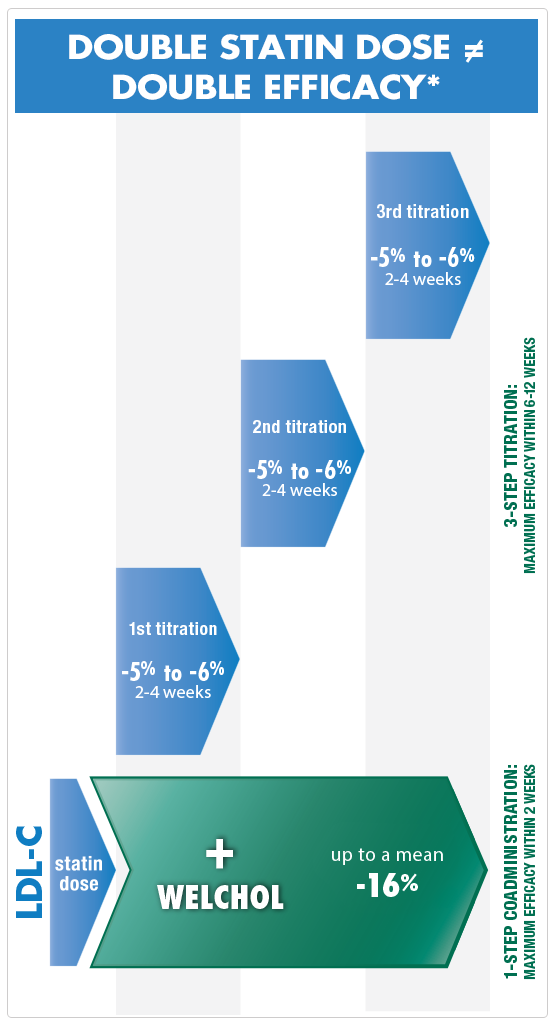
*This chart is not the result of a head-to-head clinical study, but is based on the "Rule of Six" principle: for each doubling of the statin dose, only an additional 6% further lowering of LDL-C is achieved.
Please see Important Safety Information about Welchol below.
Welchol Savings for Your Patients
Based on a $0 co-pay for a 90-day supply or a $10 co-pay for a 30-day supply. Restrictions apply based on eligibility. See Terms & Conditions below.
PRINT SAVINGSCARD NOW

Based on a $0 co-pay for a 90-day supply or a $10 co-pay for a 30-day supply. Restrictions apply based on eligibility. See Terms & Conditions below.
PRINT SAVINGSCARD NOW
Welchol is indicated as an adjunct to diet and exercise to:
- reduce elevated low-density lipoprotein cholesterol (LDL-C) in patients with primary hyperlipidemia
- reduce LDL-C levels in boys and postmenarchal girls, 10 to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH)
- improve glycemic control in adults with type 2 diabetes mellitus
- Welchol should not be used for the treatment of type 1 diabetes or for the treatment of diabetic ketoacidosis
- The effect of Welchol on cardiovascular morbidity and mortality has not been determined.
- Welchol has not been studied in type 2 diabetes in combination with a dipeptidyl peptidase-4 inhibitor
- Welchol has not been studied in Fredrickson Type I, III, IV, and V dyslipidemias
- Welchol has not been studied in children younger than 10 years of age or in premenarchal girls
To the Patient: You must present this card to the pharmacist along with your WELCHOL® (colesevelam HCl) prescription to participate in the program. For patients with commercial insurance, savings per prescription of WELCHOL will apply after the following out‐of‐pocket expenses are met: $10 per prescription for a 30‐day supply of WELCHOL or $0 per prescription for a 90‐day supply of WELCHOL. Offer may not be combined with any other program offer or discount for WELCHOL. Savings for WELCHOL are subject to a maximum benefit of $150 per 30‐day prescription or $450 per 90‐day prescription. If you have questions regarding your eligibility or benefits, or wish to discontinue participation, call (877) 264‐2440
(8 AM – 8 PM ET, Monday‐Friday). When you use this card, you are certifying that you understand the program rules, regulations, and terms and conditions. You are not eligible if you are enrolled in any state or federal health care program, including, but not limited to, Medicare Part D or Medicaid, VA, DOD, or TRICARE/CHAMPUS; or where taxed, restricted, or prohibited by law; or if you do not otherwise comply with the terms of this card. Further, you agree to discontinue using the card if you enroll in any state or federal health care program during the program period. Offer valid in US and Puerto Rico only.
To the Pharmacist: When you use this card, you are certifying that the patient is not enrolled in any federal, state, or other governmental programs for this prescription.
- Submit transaction to McKesson Corporation, using BIN #610524.
- If primary coverage exists, input card information as secondary coverage and transmit using the COB segment of NCPDP transaction. Applicable discounts will be displayed in the transaction response.
- Acceptance of this card is subject to LoyaltyScript® program Terms and Conditions posted at www.mckesson.com/mprstnc.
- Patient not eligible if enrolled in any state or federal health care program, including, but not limited to, Medicare Part D or Medicaid, VA, DOD, or TRICARE/CHAMPUS, or where taxed, restricted, or prohibited by law. Offer valid in US and Puerto Rico only.
- The LoyaltyScript® card is not valid for use with any other prescription drug discount or cash cards for WELCHOL. Claims submitted utilizing the program are subject to audit or validation.
- LoyaltyScript® is not an insurance card.
Cosette Pharmaceuticals, Inc., reserves the right to rescind, revoke, or amend this program, at any time, without notice.
Trademarks not owned by Cosette Pharmaceuticals, Inc., are property of their respective owners.
REFERENCES:
1. Illingworth DR. Management of hypercholesterolemia. Med Clin North Am 2000;84(1):23–42.
2. US Food and Drug Administration. FDA Drug Safety Communication: New restrictions, contraindications, and dose limitations for Zocor (simvastatin) to reduce the risk of muscle injury. http://www.fda.gov/Drugs/DrugSafety/ucm256581.htm. Published June 8, 2011. Accessed June 18, 2019.
3. Welchol (colesevelam HCI). Prescribing Information. Cosette Pharmaceuticals, Inc., South Plainfield, NJ; 2019.
4. Bays H, Dujovne C. Colesevelam HCl: a non-systemic lipid altering drug. Expert Opin Pharmacother. 2003;4(5):779-790.
5. Hunninghake D, Insull W Jr, Toth P, et al. Coadministration of colesevelam hydrochloride with atorvastatin lowers LDL cholesterol additively. Atherosclerosis. 2001;158:407-416.
6. Knapp HH, Schrott H, Ma P, et al. Efficacy and safety of combination simvastatin and colesevelam in patients with primary hypercholesterolemia. Am J Med. 2001;110(5):352-360.
7. Davidson M, Toth P, Weiss S, et al. Low-dose combination therapy with colesevelam hydrochloride and lovastatin effectively decreases low-density lipoproteain cholesterol in patients with primary hypercholesterolemia. Clin Cardiol. 2001;24:467-474.
© 2022 Cosette Pharmaceuticals, Inc. CP-US-WC-004 06/22

