Patient Savings

 PRINT SAVING
PRINT SAVINGCARD NOW
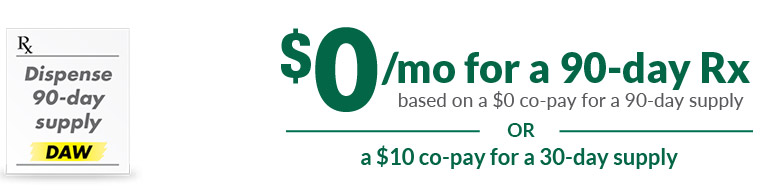
Savings for Welchol are subject to a maximum benefit of $150 for a 30-day prescription or $450 for a 90-day prescription. Offer not valid for patients enrolled in a state or federal healthcare program including, but not limited to, Medicare Part D or Medicaid, VA, DOD, or TRICARE/CHAMPUS. Other restrictions apply.
Welchol Savings for Your Patients
Based on a $0 co-pay for a 90-day supply or a $10 co-pay for a 30-day supply. Restrictions apply based on eligibility. See Terms & Conditions below.
PRINT SAVINGSCARD NOW

Based on a $0 co-pay for a 90-day supply or a $10 co-pay for a 30-day supply. Restrictions apply based on eligibility. See Terms & Conditions below.
PRINT SAVINGSCARD NOW
Welchol is indicated as an adjunct to diet and exercise to:
- reduce elevated low-density lipoprotein cholesterol (LDL-C) in patients with primary hyperlipidemia
- reduce LDL-C levels in boys and postmenarchal girls, 10 to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH)
- improve glycemic control in adults with type 2 diabetes mellitus
- Welchol should not be used for the treatment of type 1 diabetes or for the treatment of diabetic ketoacidosis
- The effect of Welchol on cardiovascular morbidity and mortality has not been determined.
- Welchol has not been studied in type 2 diabetes in combination with a dipeptidyl peptidase-4 inhibitor
- Welchol has not been studied in Fredrickson Type I, III, IV, and V dyslipidemias
- Welchol has not been studied in children younger than 10 years of age or in premenarchal girls
To the Patient: You must present this card to the pharmacist along with your WELCHOL® (colesevelam HCl) prescription to participate in the program. For patients with commercial insurance, savings per prescription of WELCHOL will apply after the following out‐of‐pocket expenses are met: $10 per prescription for a 30‐day supply of WELCHOL or $0 per prescription for a 90‐day supply of WELCHOL. Offer may not be combined with any other program offer or discount for WELCHOL. Savings for WELCHOL are subject to a maximum benefit of $150 per 30‐day prescription or $450 per 90‐day prescription. If you have questions regarding your eligibility or benefits, or wish to discontinue participation, call (877) 264‐2440
(8 AM – 8 PM ET, Monday‐Friday). When you use this card, you are certifying that you understand the program rules, regulations, and terms and conditions. You are not eligible if you are enrolled in any state or federal health care program, including, but not limited to, Medicare Part D or Medicaid, VA, DOD, or TRICARE/CHAMPUS; or where taxed, restricted, or prohibited by law; or if you do not otherwise comply with the terms of this card. Further, you agree to discontinue using the card if you enroll in any state or federal health care program during the program period. Offer valid in US and Puerto Rico only.
To the Pharmacist: When you use this card, you are certifying that the patient is not enrolled in any federal, state, or other governmental programs for this prescription.
- Submit transaction to McKesson Corporation, using BIN #610524.
- If primary coverage exists, input card information as secondary coverage and transmit using the COB segment of NCPDP transaction. Applicable discounts will be displayed in the transaction response.
- Acceptance of this card is subject to LoyaltyScript® program Terms and Conditions posted at www.mckesson.com/mprstnc.
- Patient not eligible if enrolled in any state or federal health care program, including, but not limited to, Medicare Part D or Medicaid, VA, DOD, or TRICARE/CHAMPUS, or where taxed, restricted, or prohibited by law. Offer valid in US and Puerto Rico only.
- The LoyaltyScript® card is not valid for use with any other prescription drug discount or cash cards for WELCHOL. Claims submitted utilizing the program are subject to audit or validation.
- LoyaltyScript® is not an insurance card.
Cosette Pharmaceuticals, Inc., reserves the right to rescind, revoke, or amend this program, at any time, without notice.
Trademarks not owned by Cosette Pharmaceuticals, Inc., are property of their respective owners.
© 2022 Cosette Pharmaceuticals, Inc. CP-US-WC-004 06/22

